Benefit from our experience – we take the pressure off your daily routines.
We at SITEWORKS are a team of professionals, all with long-time experience in clinical research. The sector is already highly complex and specialized, aspects that are intensifying daily. This makes it increasingly difficult for research-savvy doctors to reconcile a role in research & development with their day-to-day work under Germany’s healthcare system.
Do you share our passion & motivation to assume a role in research and development?
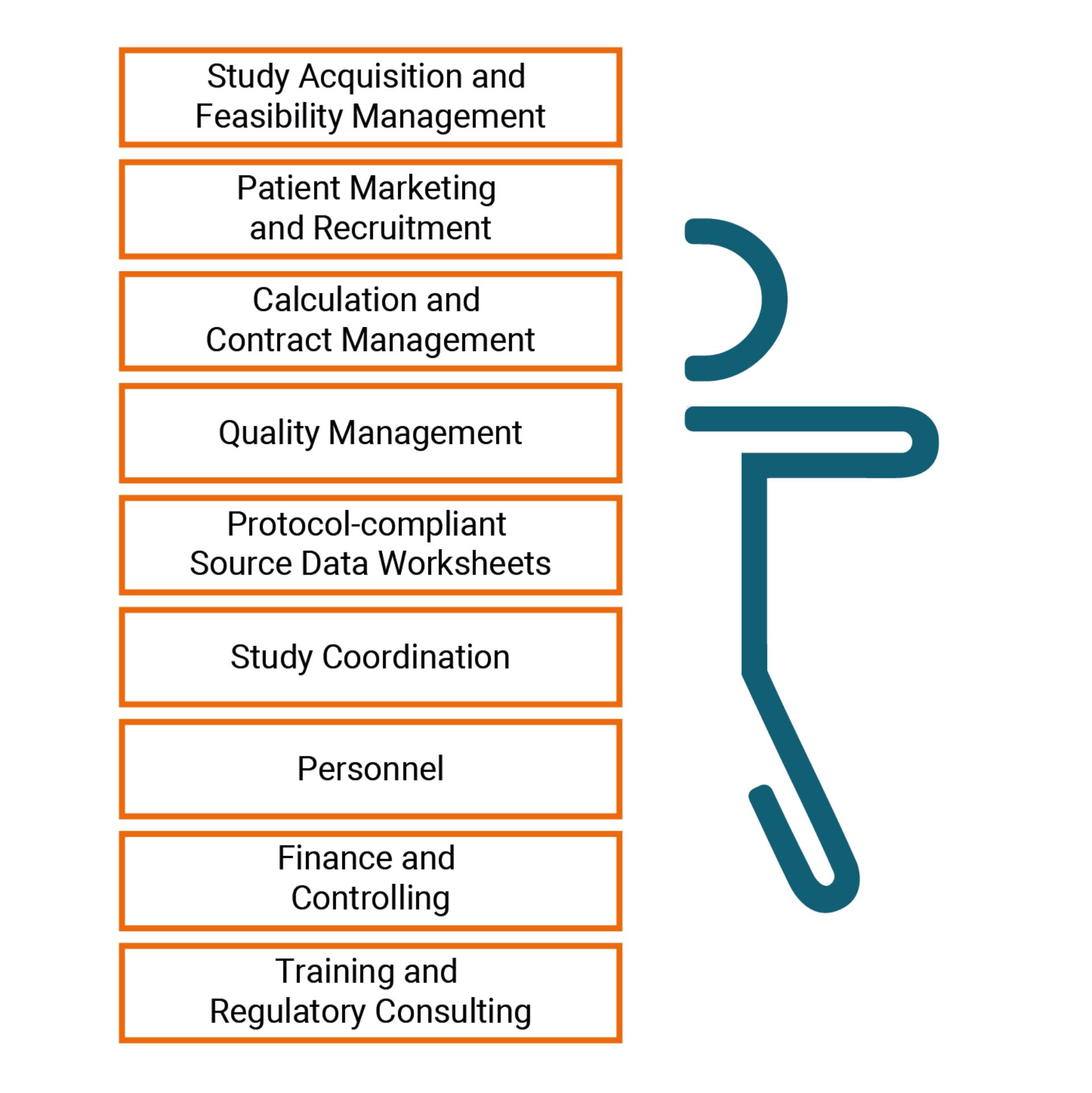
We provide support for all aspects of study execution – on the administration, regulation, marketing, organization and staffing sides – enabling you to combine clinical trial execution with your day-to-day routines in standard medical care.
We will take work off your shoulders and create the space you need to focus on the essentials while ensuring that your trial is conducted to binding protocols, namely, on your patients and your medical work. Why not join our network and leave the organization to us?
We offer a customized service to meet your individual requirements – what is it that you need?
Optimal performance and high-quality data reflect a highly professional approach. This ensures that clients continue to opt for specific research sites over the long term – now and in the future.
Research sites give you access to cutting-edge medical therapies and alternative treatment for your patients without putting extra strain on your budget. Patients themselves remain in their home environments – it is vitally important for us to maintain this bond of trust.
- Trial acquisition and market access? We take responsibility.
- Study personnel? We provide flexible staffing capacities with clinical trial experience.
- Study organization? For us, optimal coordination is standard business practice. We provide swift, flexible support for physicians who cooperate with us - from feasibility studies, contract management and patient recruitment to handling outpatient appointments, documentation and invoicing.
- Trial quality? No need to worry about growing protocol complexity and the stringent requirements placed on us as a result by our clients. We are familiar with many different designs and the processes involved, and work to a well-established quality management system. Audits and inspections are part and parcel of our day-to-work – no problem with a sound underlying base!